Search results
Search for "structural modification" in Full Text gives 38 result(s) in Beilstein Journal of Organic Chemistry.
N-Boc-α-diazo glutarimide as efficient reagent for assembling N-heterocycle-glutarimide diads via Rh(II)-catalyzed N–H insertion reaction
Beilstein J. Org. Chem. 2023, 19, 1841–1848, doi:10.3762/bjoc.19.136
- circumstances, considerably impacting their pharmacological utility [19][20], Moreover, the conventional CRBN recruiters restrict structural modification options necessary to maintain satisfactory affinity for the E3-ligase [21][22][23][24]. These limitations underscore the relevance of expanding the chemical
Quinoxaline derivatives as attractive electron-transporting materials
Beilstein J. Org. Chem. 2023, 19, 1694–1712, doi:10.3762/bjoc.19.124

- of structural modification, the introduction of various functional groups into Qx derivatives has allowed for the precise control of energy levels, bandgaps, and carrier transport properties. For instance, Sharma et al. fine-tuned the intermolecular charge transfer (ICT) transitions and emission
Synthesis and biological evaluation of Argemone mexicana-inspired antimicrobials
Beilstein J. Org. Chem. 2023, 19, 1511–1524, doi:10.3762/bjoc.19.108
- structural modification, this was chosen as the path towards enhancing the activity of B1. As shown in Scheme 5, we reduced B1 to produce B14, which was then screened against our panel of microbial organisms (Table 2). We were very pleased with the results of B14, representing a near universal improvement
Synthesis of ether lipids: natural compounds and analogues
Beilstein J. Org. Chem. 2023, 19, 1299–1369, doi:10.3762/bjoc.19.96
- installation of the phosphocholine moiety (67%) followed by the deprotection of the secondary alcohol (100%) and its acetylation (53%) produced 13.6. Wissner et al. also reported the incorporation of a gem-dimethyl substituent on the glycerol backbone [97]. One illustration of this structural modification is
- alcohol produced 27.6 that, in a two-step sequence, was used to install the phosphocholine moiety to produce 27.8. The double structural modification of edelfosine that consists to link the lipid chain via a thioether function and to replace the phosphate moiety by a thiophosphate was reported by
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
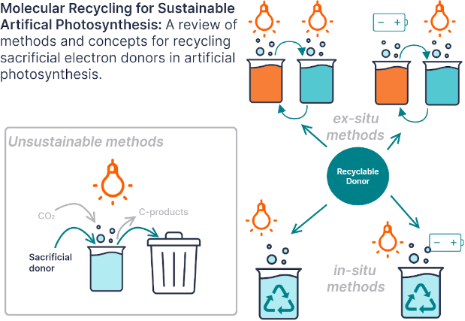
- mediators that can be recycled photo- and electrochemically [45][49]. As illustrated in Figure 5, structural modification of the benzimidazole core alters the redox behavior and allows tuning of the oxidation potential. The benzimidazoles shown all have enough reducing power to reductively quench Ru(bpy)3
Exploring the role of halogen bonding in iodonium ylides: insights into unexpected reactivity and reaction control
Beilstein J. Org. Chem. 2023, 19, 1171–1190, doi:10.3762/bjoc.19.86

- hard fluoride interacts with the stronger σ-hole. Structural modification of the β-dicarbonyl auxiliary has led to improved outcomes, due to increased ylide stability rather than decreasing activation energies of the fluorination reaction. There are, however, other conflicting pieces of evidence that
The asymmetric Henry reaction as synthetic tool for the preparation of the drugs linezolid and rivaroxaban
Beilstein J. Org. Chem. 2022, 18, 438–445, doi:10.3762/bjoc.18.46
- 15 and 19 was also performed with the aim to increase the enantiomeric purity of the corresponding nitroaldol products 21–26. The structural modification consisted in the introduction of different alkyl moieties to the carbamate functional group of the aldehyde intermediates 15–20. As bulky and/or
- 2). The bulky (R2 = t-Bu) or chiral (R2 = ʟ-menthyl or (−)-bornyl) alkoxy groups (derived from relatively inexpensive and readily available alcohols) were introduced into the carbamate moiety instead of an ethyl group as in aldehydes 15 and 19. Here, the influence of this structural modification of
Synthetic strategies toward 1,3-oxathiolane nucleoside analogues
Beilstein J. Org. Chem. 2021, 17, 2680–2715, doi:10.3762/bjoc.17.182
On the application of 3d metals for C–H activation toward bioactive compounds: The key step for the synthesis of silver bullets
Beilstein J. Org. Chem. 2021, 17, 1849–1938, doi:10.3762/bjoc.17.126

Distinctive reactivity of N-benzylidene-[1,1'-biphenyl]-2-amines under photoredox conditions
Beilstein J. Org. Chem. 2020, 16, 1335–1342, doi:10.3762/bjoc.16.114
- hypothesized that the structural modification of the substrate might affect its reactivity, and to our delight, the introduction of an ortho-phenyl moiety on the N-benzylideneaniline species provided the desired 1,2-diamine product, wherein the N,N-dicyclohexylmethylamine (Cy2NMe) acted as both the coupling
Synthesis, antiinflammatory activity, and molecular docking studies of bisphosphonic esters as potential MMP-8 and MMP-9 inhibitors
Beilstein J. Org. Chem. 2020, 16, 1277–1287, doi:10.3762/bjoc.16.108
- approximation, to study the effect of these structural modification on the pharmacodynamics, we performed a molecular docking over two MPPs. In Table 4, the interaction energy value (MolDock Score) [38] of each compound with the two different MMPs obtained from the docking calculation is displayed. Also, the
Development of fluorinated benzils and bisbenzils as room-temperature phosphorescent molecules
Beilstein J. Org. Chem. 2020, 16, 1154–1162, doi:10.3762/bjoc.16.102
- (Figure 2A), which show prominent fluorescence not only in dilute solution, but also in the crystalline state [27][28][29][30][31]. As a powerful tool to develop novel pure organic phosphorescent molecules, we envisioned the structural modification of the carbon–carbon triple (C≡C) bond in fluorinated
- effects of structural modification (i.e., the benzil structure with a tolane vs bisbenzil moiety) and incorporation of fluorine atoms on the phosphorescence, the ratio between the peak intensities at ≈395 and ≈560 nm (I560/I395) was quantitatively calculated, and the results are summarized in Table 1. The
Synthesis of 9-O-arylated berberines via copper-catalyzed CAr–O coupling reactions
Beilstein J. Org. Chem. 2019, 15, 1575–1580, doi:10.3762/bjoc.15.161
- 9-O-phenylene-bridged berberine dimer (5) was synthesized by copper-catalyzed cross-coupling of tetrahydroberberrubine and aryl iodides, followed by oxidation with I2. Keywords: arylation; berberines; cross-coupling; copper; lipophilicity; structural modification; Introduction Berberine (BBR) is a
Chemical structure of cichorinotoxin, a cyclic lipodepsipeptide that is produced by Pseudomonas cichorii and causes varnish spots on lettuce
Beilstein J. Org. Chem. 2019, 15, 299–309, doi:10.3762/bjoc.15.27
- treatment had no activity, supporting that a macrocyclic peptide would be essential for biological activity. The sequence Z-dhThr17-D-alloThr18 of cichorinotoxin was converted to Z-dhThr17-E-dhThr18 by alkaline hydrolysis. This additional structural modification may have further decreased the activity
Semi-synthesis and insecticidal activity of spinetoram J and its D-forosamine replacement analogues
Beilstein J. Org. Chem. 2018, 14, 2321–2330, doi:10.3762/bjoc.14.207
- as the glycosylation donor of C9–OH, but also the protecting group of C17–OH, greatly reducing the synthetic steps and costs. Macrolide compounds are a new kind of insecticides and fungicides which have also been widely applied in medicine [14][15]. Currently, research on structural modification of
Functionalization of graphene: does the organic chemistry matter?
Beilstein J. Org. Chem. 2018, 14, 2018–2026, doi:10.3762/bjoc.14.177
- effects and to open new avenues for the application of GO and RGO, a chemical functionalization was conducted [9][10][11][12]. There are two approaches for the structural modification of GO and RGO: (i) the reaction of oxygen-bearing groups or (ii) the functionalization of sp2 carbons from the graphene
- functionalization of the graphene-family material. In fact, the structural modification of GO and RGO constitutes a key starting point of such research. Even though many studies on chemical functionalization have presented very interesting and novel applications of GO and RGO derivatives they also present yet
Recent advances in phosphorescent platinum complexes for organic light-emitting diodes
Beilstein J. Org. Chem. 2018, 14, 1459–1481, doi:10.3762/bjoc.14.124

- between hundreds of nanoseconds to several microseconds, constituting a formally spin-forbidden transition (phosphorescence). Structural modification of the TMCs and proper tailoring of coordinated ligands can independently act on the nature, energy and topology of frontier orbitals. In fact, a fine
Synthesis of fluoro-functionalized diaryl-λ3-iodonium salts and their cytotoxicity against human lymphoma U937 cells
Beilstein J. Org. Chem. 2018, 14, 364–372, doi:10.3762/bjoc.14.24
- suggested that antitumor drug candidates could be designed by further structural modification of these compounds 3. Moreover, with 3p exhibiting the greatest potency against U937 cells with comparably lower toxicity against AGLCL cells, further biological studies using 3p including in vivo evaluation should
Mechanochemical synthesis of thioureas, ureas and guanidines
Beilstein J. Org. Chem. 2017, 13, 1828–1849, doi:10.3762/bjoc.13.178

- Cs2CO3 for 3 hours finally afforded phenytoin in an excellent 84% isolated yield (Scheme 16c). The introduction of a sulfonyl group on the urea framework has been found to be the crucial structural modification in the development of the 1st generation antidiabetic drugs such as tolbutamide and
Synthesis of acylhydrazino-peptomers, a new class of peptidomimetics, by consecutive Ugi and hydrazino-Ugi reactions
Beilstein J. Org. Chem. 2016, 12, 2865–2872, doi:10.3762/bjoc.12.285
- peptidomimetics with potential biological activity. Keywords: acylhydrazino-peptomers; consecutive Ugi reactions; peptide-peptoid hybrid; peptomer; Introduction In the last decades, increasing efforts have been extensively carried out to improve the pharmacological properties of natural peptides by structural
- modification of the amino acids [1][2][3][4][5][6][7][8]. These modifications allowed the obtention of molecules that mimic the properties of peptides (peptidomimetics) but usually exhibit greater proteolytic stability, increased cellular permeabilities and avoid stereochemical constraints. Figure 1 represents
Towards the development of continuous, organocatalytic, and stereoselective reactions in deep eutectic solvents
Beilstein J. Org. Chem. 2016, 12, 2620–2626, doi:10.3762/bjoc.12.258
- advantages in terms of reaction sustainability. In particular, the possibility to strongly reduce the amounts of organic solvent and the recyclability of the catalyst were demonstrated [23]. Moreover, in this approach, no structural modification of the precious chiral catalyst was necessary. A well-explored
Effects of solvent additive on “s-shaped” curves in solution-processed small molecule solar cells
Beilstein J. Org. Chem. 2016, 12, 2543–2555, doi:10.3762/bjoc.12.249

- . The structural modification had the desired effect of reducing the band gap for extended absorption in the visible spectrum while maintaining a low-lying HOMO level to achieve high VOC. The energy levels are nearly ideal match for incorporation into BHJ devices with the acceptor PC71BM, maximizing
Synthesis and characterization of benzodithiophene and benzotriazole-based polymers for photovoltaic applications
Beilstein J. Org. Chem. 2016, 12, 1629–1637, doi:10.3762/bjoc.12.160
- , the BDT monomers used for the synthesis of PTzBDT-1 and PTzBDT-2 were substituted with either 2-octylthienyl (BDT-1) or 2,3-dihexylthienyl (BDT-2) as side groups, respectively. As a consequence of this fine structural modification on the BDT moiety, useful information on the effect of the different
Muraymycin nucleoside-peptide antibiotics: uridine-derived natural products as lead structures for the development of novel antibacterial agents
Beilstein J. Org. Chem. 2016, 12, 769–795, doi:10.3762/bjoc.12.77
- search for completely unknown structures. For such novel agents, natural products appear to be a promising source [7][8][9]. Bacteria deploy different mechanisms to escape the toxic effect of an antibacterial drug [10][11][12]. These include the structural modification and degradation of a drug, as it is
Unconventional application of the Mitsunobu reaction: Selective flavonolignan dehydration yielding hydnocarpins
Beilstein J. Org. Chem. 2016, 12, 662–669, doi:10.3762/bjoc.12.66
- silymarin complex for protection of liver damage, and for preventing skin tumor promotion, these compounds have attracted efforts on their total synthesis (e.g., isosilybin) [6] and structural modification [5][7][8][9] to improve their water-solubility, bioavailability and biological activities. Recently

















































































































































































































































































































































































































































































































